Conflicts and their resolution are not uncommon in science. This essay examines conflicting ideas regarding cellular homeostasis related to cytoplasmic and mitochondrial calcium.
In this, I played a minor role by testing how mitochondria affect cellular calcium balance in the giant neuron of a marine worm.
Early in my career, the giant neuron of an invertebrate marine annelid, Myxicola infundibulum, was my chosen experimental preparation. In Myxicola cytoplasm, a large mitochondrial component for calcium homeostasis was found. This was not a particularly welcome observation among some, as I found out when I began trying to publish. Mitochondrial calcium was, at this time, considered to have a role in regulating mitochondrial matrix enzymes, but its only role regarding cytoplasmic calcium homeostasis was thought to be at the end of a cell’s life, contributing to necrotic cell death.
This perspective continued even after my research career ended in 2011. This is reflected in a 2016 review with the following quotation 1: “Evidence suggests that an increase in mitochondrial calcium can augment ATP production by altering the activity of calcium-sensitive mitochondrial matrix enzymes. In contrast, the entry of large amounts of mitochondrial calcium in the setting of ischemia-reperfusion injury is thought to be a critical event in triggering cellular necrosis.”
In the section on the Physiological Role of Mitochondrial Calcium1, is the additional quotation: “Studies in the mid-1970s suggested that mitochondrial calcium might serve to regulate or modulate cytosolic calcium or act as a source for calcium release by a physiological agonist.2,1 However, the latter hypothesis became unlikely when it became apparent that the endoplasmic reticulum/sarcoplasmic reticulum is the primary source of agonist-induced calcium release.”
Though a mitochondrial role in physiological calcium uptake was out of favor for some decades after the 1980s, by 2000, other opinions were emerging 3,1. Even in the review of 2016 there was an earlier cited result that mitochondria may be able to import calcium at very low (physiological) concentrations: “For example, when rat liver mitochondria are pulsed for a few seconds with nanomolar levels of calcium, they exhibit calcium uptake with a rapid kinetics which was termed rapid mode calcium uptake.4,5”
The prevailing view, however, had been that “Given the measured low affinity (≈ 5–10 μmol/L) for (mitochondrial) calcium uptake, it initially seemed that under physiological conditions, cytosolic calcium would never rise to levels high enough to activate the MCU (Mitochondrial Calcium Uniporter).” But the proviso was added: “However, it was later demonstrated that microdomains of high calcium can exist near the junction of mitochondria and the site of calcium release (the endoplasmic/sarcoplasmic reticulum) and that the concentration of calcium in these microdomains is, in fact, high enough to allow calcium uptake via the (mitochondrial) uniporter (MCU).”6,1
From this scholarly review1 is the statement: “Measurements of basal cytosolic calcium were thought to be in the range of 100 nmol/L and peak calcium transients were <1 μmol/L.7,8 Given the measured low affinity (≈5–10 μmol/L) for calcium uptake, it initially seemed that under physiological conditions, cytosolic calcium would never rise to levels high enough to activate the MCU.”
In conclusion, mitochondria were generally believed be of little use for physiological cytosolic calcium regulation, though there were emerging doubts about this.
A review of 2024, only eight years later, expresses with more certainty that mitochondria do have an important physiological role in this important function.9
Statement from this review 9: “In this context, mitochondria play a pivotal role. Indeed, in response to an increase in [Ca2+]cyt, mitochondria can uptake Ca2+ through a process that depends on three prerequisites.
“1) The electrochemical proton gradient (ΔμH+), generated by the translocation of H+ ions across the inner mitochondrial membrane (IMM) due to the activity of the electron transport chain …10: It comprises the membrane potential difference (ΔΨ) and the H+ concentration difference (ΔpH), with ΔΨ being predominant. The negative ΔΨ (~-180 mV) represents the driving force for Ca2+ accumulation within the mitochondrial matrix.
“2) The microdomains between ER/SR and the plasma membrane with the mitochondria: Mitochondria are closely located to ER/SR Ca2+ channels (inositol-1,4,5-triphosphate-receptors [Ins(1,4,5)P3Rs] and ryanodine receptors (RYRs), as well as plasma membrane Ca2+ channels (store-operated channels and voltage-operate channels). This enables mitochondria to promptly sense microdomains of high [Ca2+]cyt 11,12,13.
“3) The presence of the mitochondrial Ca2+ uniporter (MCU) complex in the IMM, a sophisticated mechanism that enables Ca2+ entry into the mitochondrial matrix: … the presence of microdomains between mitochondria and ER/SR is an essential prerequisite for mitCa2+ uptake. Rapid changes in [Ca2+]cyt occurring at these sites provide regulatory feedback on ER/SR Ca2+ channels.”
The presence of microdomains of elevated calcium and calcium uptake by mitochondria under physiological conditions by 2024 seemed well established. 13,14.
Personal reflections:
Even though opinions evolved and these currents of thought converged to a general scientific consensus, strong opinions and personalities had posed fertile ground for disagreements. Early in my career(1980s), I had been collecting data and attempting to report my findings, which showed different factors influencing mitochondrial calcium uptake. To some well-known scientists at the time , these measurements seemed unimportant at best and misleading at worst. They were thinking that mitochondrial calcium had an insignificant physiological role for cellular calcium regulation, and many showed me that they were unhappy with my results.
Here is an example: In 1986, my department chairman relayed a message that a national leader in this field had admonished him not to ask for a letter of recommendation when the time came for a decision about my tenure. Individual egos can have a considerable impact within academic settings, especially when differing viewpoints concerning research findings are involved.
Here, I summarize some Myxicola findings, which align closely with current perspectives on mitochondrial calcium’s role.
A. In neuronal cytoplasm from Myxicola, measurements with calcium-sensitive and pH-sensitive electrodes produced the following results, eventually published, in 1986:15
1. Where mitochondria are concentrated, greater calcium buffering may be seen.
2. Electron microscopic images show mitochondria and ER-type intracellular membranes juxtapositioned within the cytoplasm.
3 . The energy dependence of calcium buffering suggests a strong mitochondrial component.
4. Adding hydrogen ions (lowering the pH) causes free calcium in the cytoplasm to increase.
5. Elevated free calcium in the cytoplasm has a small, but measurable, effect in elevating the hydrogen ion concentration.
6. In dialyzed axoplasm with a free calcium concentration well below the μmolar concentration range, mitochondria clearly lowered ambient free calcium.
B. Cytoplasmic calcium uptake was also measured using radioactive calcium (45Ca), and the following properties were published in1993: 16
1. 45Ca uptake by Myxicola axoplasm was strongly dependent on the free calcium concentration and pH.
2. The mitochondrial uptake of TPP+, a membrane-permeant cation used to assess membrane potential, shares similar pH dependence with calcium uptake, suggesting both follow comparable mechanisms.
3. Mitochondrial inhibitors inhibited both these processes.
4. Myxicola cytoplasm was tested at three concentrations of free calcium with qualitatively similar Ca uptake results for each. A calcium concentration of 0.5 μmolar is within the physiological range.
5. The use of pharmacological agents, targeting either the endoplasmic reticulum or mitochondria, demonstrated that both components are present.
6. However, approximately 80% of the calcium uptake could be attributable to a mitochondrial mechanism, even at physiological calcium concentrations.
7. ER and mitochondrial inhibitors, when added separately and in combination, suggested a functional relationship between the mitochondrial and non-mitochondrial calcium stores in Myxicola.
I retired in 2011. In 1999, several years before, I spent a sabbatical year examining how mitochondria shape intracellular calcium signals involved in the release of reproductive hormones. These studies incorporated compartmentalized fluorescent calcium indicators in pituitary gonadotrophs. Both mitochondrial and cytoplasmic calcium were simultaneously measured.
The findings showed how mitochondria and non-mitochondrial sources each take up and release calcium, highlighting that mitochondria play a key role in controlling calcium levels inside gonadotrophs. Below is an excerpt from the introduction of the publication, demonstrating the diverse perspectives that existed at the time:
“A mitochondrial contribution to intracellular Ca2+dynamics has been debated for several decades. For some time, it was supposed that mitochondria do not accumulate significant Ca2+ unless the surrounding free cytoplasmic Ca2+ concentration ([Ca2+]cyt)is dangerously high17, 18 . This view seemed plausible because accumulating Ca2+ would divert energy of the mitochondrial membrane potential from its normal function of producing ATP, and, because the endoplasmic reticulum is already specialized for sequestering and releasing Ca2+, regulation by yet another organelle seemed unnecessary. Nevertheless, in some cells, mitochondria do take up Ca2+ at apparently physiological [Ca2+]cyt levels (<2 μm)”3,15,16
Findings from the pituitary gonadotroph supported mitochondria’s role in regulating cellular calcium levels.
Darwin and the evolutionary development of living organisms, their diversity and similarity at the cellular level: When studying biology, scientists select an organism and cell type but base their conclusions on the principle that protein structures and basic processes are generally shared across species and cytology due to evolution. However, exceptions may exist, such as appears in the giant axons found in Loligo pealii squid and the marine worm Myxicola.
The squid had long been used to study signal propagation across species. However, when it comes to intracellular calcium homeostasis, differences emerge. In Myxicola, mitochondria are key to calcium regulation, as in many other organisms. The squid giant axon appears to be an exception: research had shown that its mitochondria contribute minimally—just six percent—to calcium homeostasis7. In the 1980s, the squid results were thought to be representative of the biological norm in general.
Scientific advancement often involves complexities, but it thrives when researchers consistently gather data, reassess findings, and pursue further experiments. Although personal ambitions sometimes influence the process, time ultimately brings greater understanding. Science continually develops.
It seems reasonable to suggest now that the initial findings from the humble marine worm Myxicola offered broader insights into calcium regulation and better represented the fundamental mechanisms involved.
If this essay has one lesson from an eighty-year-old retiree to young scientists, it’s to remain honest and diligent when gathering your data. Once you have done this, stand firmly by your findings, even if you face considerable pressure to do otherwise.
A figure from reference (15):
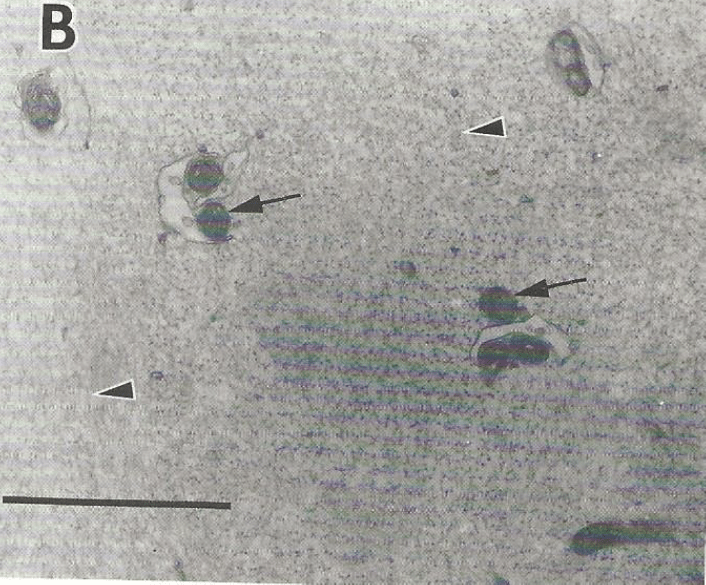
The Figure shows an electron microscopic image of compacted Myxicola axoplasm. Two of the arrows point to mitochondria, which are closely attached to vesicles that resemble the endoplasmic reticulum. The third arrow in the upper right is within a mitochondria-free region containing what are likely neurofilaments which are abundant in Myxocola axoplasm. The bar represents 1.3 μm of length.
Journal References
1. Finkel, Menazza, Holmström, Parks, Liu, Junhui Sun, Jie Liu, Xin Pan, Murphy. (2016) The Ins and Outs of Mitochondrial Calcium. Circulation Research.
2. Nicholls DG. (1978) The regulation of extramitochondrial free calcium ion concentration by rat liver mitochondria. Biochem J. 176:463–474
3. Kaftan, Xui, Abercrombie, and Hille. (2000) Mitochondria shape hormonally induced cytoplasmic calcium oscillations and modulate exocytosis. J Biol Chem 275, 33: 25465-470
4. Sparagna, Gunter, Sheu, Gunter. (1995) Mitochondrial calcium
uptake from physiological-type pulses of calcium. A description of the
rapid uptake mode. J Biol Chem. 270:27510–27515
5. Buntinas, Gunter, Sparagna, Gunter. (2001) The rapid mode of calcium uptake into heart mitochondria (RaM): comparison to RaM in liver
mitochondria. Biochim Biophys Acta. 1504:248–261
6. Rizzuto, Brini, Murgia, Pozzan. (1993) Microdomains with high Ca2+
close to IP3-sensitive channels that are sensed by neighboring mitochondria.
Science. 262:744–747
7. Brinley, Tiffert, Scarpa, Mullins. (1977) Intracellular calcium buffering capacity in isolated squid axons. J Gen Physiol. 70:355–38.
8. Allen, Blinks. (1978) Calcium transients in aequorin-injected frog cardiac muscle. Nature. (1978) 273:509–513
9. D’Angelo, Reane and Raffaello. (2023) Neither too much nor too little: mitochondrial calcium concentration as a balance between physiological and pathological conditions. Frontiers in Molecular Bioscience.
10. Mitchell and Moyle. (1967) Chemiosmotic hypotheses of oxidative phosphorylation, Nature. 213: 137-139
11. Rizzuto, Pinton, Carrington, Fay, Fogarty, Lifshitz, Tuft, Pozzan. (1998) Close contacts with the endoplasmic reticulum as determinants of mitochondrial Ca2+ responses. Science. 280(5370):1763-6
12. Csordás, Thomas, and Hajnóczky (1999). Quasi-synaptic calcium signal transmission between endoplasmic reticulum and mitochondria. EMBO J. 18, 96–108
13. Szalai, Csordás, Hantash, Thomas, and Hajnóczky. (2000) Calcium signal transmission between ryanodine receptors and mitochondria. J. Biol. Chem. 275, 15305–15313
14. Rizzuto, De Stefani, Raffaello, Mammucari. (2012) Mitochondria as sensors and regulators of calcium signalling. Nat Rev Mol Cell Biol. 13(9):566-78
15. Abercrombie and Hart. (1986) Calcium and proton buffering and diffusion in isolated cytoplasm from Myxicola axons. Am J. Physiol 250 (cell Physiol. 19) C391- c405
16. Al-Baldawi, Moore, and Abercrombie. (1993) Calcium accumulation by organelles within Myxicola axoplasm J. Physiol 461: 633-646
17. Carifoli. (1987) Ann. Rev Biochem. 56: 395-433
18. Somlyo and Himpens . (1989) FASAB J 250: C391-C405
19. Blaustein. (1988) Calcium transport and buffering in neurons. Trends in Neurosciences 11(10) 438-43
Leave a comment